Company profile
Study shows that polyphenol-rich extracts can modulate the gut microbiota conferring a cardiovascular health benefit
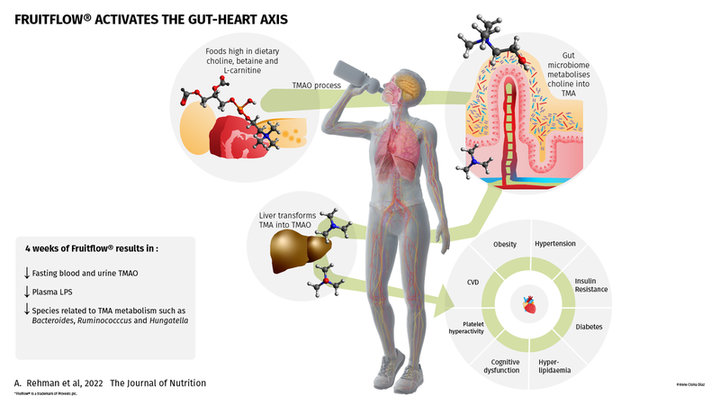
A recent clinical trial published by The Journal of Nutrition found that four weeks of supplementation with Fruitflow® (1), a water-soluble tomato concentrate, decreases fasting blood and urine TMAO when compared to baseline as well as plasma lipopolysaccharide.
The gut-heart axis
Evidence suggesting the role of the gut microbiota in maintaining host health has become mainstream. Multiple strategies to modulate the gut microbiome composition have emerged such as Probiotics, Prebiotics, Synbiotics or Faecal Microbiota Transplant.
Among the list of prebiotic fibers, plant polyphenols with an estimated 90–95% that are not absorbed in the small intestine but reach the colon, where they undergo extensive biotransformation by the gut microbiota. Dr. Robert E. Steinert, main author of this publication, explained that natural products rich in polyphenols have been hypothesized to lower plasma trimethylamine-n-oxide (TMAO), an emerging biomarker known for its proatherogenic effects by beneficially modulating the intestinal microbiota.
Dr. Marcus Claesson added: Previously thought to be a waste product of choline metabolism with no physiological function, trimethylamine-N-oxide (TMAO), a chemical metabolite derived from diet and metabolized by the gut microbiota, is now supported by growing evidence for its involvement in inflammation, cardiovascular disease, and obesity. A high level of TMAO may increase the risk of clot-related events like heart attack and stroke, according to research (2). Thus, a changed gut microbiota and increased intestinal permeability may be the causes of chronic inflammation, which worsens cardiac function (3).
TMA is a microbial metabolite produced by the gut microbiome from dietary phosphatidylcholine and L-carnitine, commonly found in red meat, cheese and eggs. When absorbed by the intestinal epithelium and transported to the liver, TMA is transformed into TMAO. TMAO is known for its proinflammatory and proatherogenic activities. Higher levels of TMAO have been linked to major adverse cardiovascular events.
Evidence suggesting the role of the gut microbiota in maintaining host health has become mainstream. Multiple strategies to modulate the gut microbiome composition have emerged such as Probiotics, Prebiotics, Synbiotics or Faecal Microbiota Transplant.
Among the list of prebiotic fibers, plant polyphenols with an estimated 90–95% that are not absorbed in the small intestine but reach the colon, where they undergo extensive biotransformation by the gut microbiota. Dr. Robert E. Steinert, main author of this publication, explained that natural products rich in polyphenols have been hypothesized to lower plasma trimethylamine-n-oxide (TMAO), an emerging biomarker known for its proatherogenic effects by beneficially modulating the intestinal microbiota.

Dr. Marcus Claesson added: Previously thought to be a waste product of choline metabolism with no physiological function, trimethylamine-N-oxide (TMAO), a chemical metabolite derived from diet and metabolized by the gut microbiota, is now supported by growing evidence for its involvement in inflammation, cardiovascular disease, and obesity. A high level of TMAO may increase the risk of clot-related events like heart attack and stroke, according to research (2). Thus, a changed gut microbiota and increased intestinal permeability may be the causes of chronic inflammation, which worsens cardiac function (3).

TMA is a microbial metabolite produced by the gut microbiome from dietary phosphatidylcholine and L-carnitine, commonly found in red meat, cheese and eggs. When absorbed by the intestinal epithelium and transported to the liver, TMA is transformed into TMAO. TMAO is known for its proinflammatory and proatherogenic activities. Higher levels of TMAO have been linked to major adverse cardiovascular events.
Maritech® fucoidan is the world’s only high purity, certified organic fucoidan with global regulatory acceptance – including FDA notified GRAS and EU Novel Foods Approval.
The research team
The research team lead by Dr. Steinert, Principal Scientist at DSM and University Lecturer at Zurich University, hypothesized that Fruitflow®, a water soluble tomato concentrate, rich in secondary metabolites, including nucleosides, phenolic conjugates, and polyphenols, would lower TMAO levels by modulating gut microbiota.
Fruitflow® is well-known for its cardiovascular benefits; several clinical trials (4)document reduced platelet aggregation, which contributes to healthy blood flow. In fact, it was the first ingredient in achieving a positive EFSA opinion and an approved innovative health claim by the European Commission (Article 13.5 claim).
Dr. Steinert explained that Atlantia Clinical Trials Ltd., was contracted by DSM to carry out the clinical trial, as they were interested in investigating whether Fruitflow® can be a dietary supplement solution targeting the gut-heart axis.
Introducing DSM’s Health from the Gut (HfG), a redefined health benefit platform for dietary supplement customers. Leading the way with an innovative range of purpose-led solutions that enhance gut health to support immunity, digestion, brain health, stress management, metabolic health and more. The aim is to serve as an end-to-end purpose-led partner that helps our customers to stay ahead of consumer demands and market needs with an agile, full-service business model that delivers standout dietary supplement solutions.
Microbiome analysis were performed out by at SeqBiome Ltd. Dr. Claesson explained the work provided by his team: SeqBiome carried out microbiome analysis of the 16S rRNA amplicon sequence data from the collected stool samples using state-of-the-art bioinformatics methods, followed by interpretation and production of publication-ready figures and tables. Analysis of clinical outcomes were carried out by the team lead by Gillian DunnGalvin at Atlantia Clinical Trials Ltd. as highlighted by Dr. DunnGalvin: Atlantia provided an end-to-end service from protocol development to support in publication writing. The statistical team completed the analysis of the clinical outcomes of urine TMAO and plasma TMAO, LPS, hsCRP and tolerability.

About this study
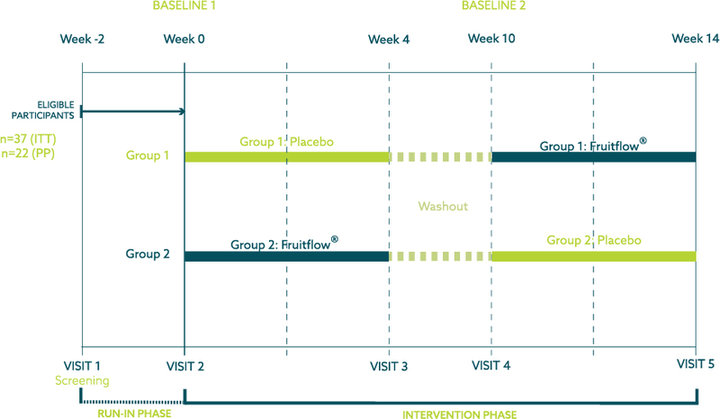
A randomized double-blind, placebo-controlled crossover study was conducted in order to assess the research question. Consisting of five visits and six-week washout, the study lasted 14 weeks after a 21-day run-in period.
The study population consisted of 40 healthy, overweight, and obese adults (BMI 28-35 kg/m2), aged 35-65 years. 20 participants were allocated to the Placebo/Fruitflow® arm (group 1), and 20 participants to the Fruitflow®/Placebo arm (group 2). Three participants self-withdrew after randomisation, the remaining 37 participants completed the study as planned and analysed in the Intention-to-Treat population. All participants were evaluated on a case-by-case basis by an independent committee before unblinding of the data to determine inclusion to the per-protocol population. 15 subjects were excluded from the Per Protocol subset analysis due to intake of antibiotic concomitant medications and/or product intake compliance below 80% which would impact the efficacy assessment of microbiome outcomes. Blood, urine and faecal samples were collected in this study. A venous blood sample was collected for safety profiling and analysis of plasma TMAO and lipopolysaccharides (LPS). Urine sample was collected for analysis of urine TMAO. Faecal samples were utilised to analyse the microbiome composition.
In addition to biological samples, questionnaires such a Gastrointestinal Symptom Rating Scale (GSRS) and Bristol Stool Chart were completed to assess tolerability. A Food Frequency Questionnaire (FFQ) was completed by the participants to determine stability of dietary intake over time.
In addition to the main intervention a subgroup of nine participants completed an acute egg challenge. They were instructed to follow a standardised consisted of a high-yolk scrambled egg recipe. The goal of this challenge test was to determine the effect of intervention on postprandial plasma and urine TMAO concentrations.
The investigational product, Fruitflow®, was commercially produced by DSM Nutritional Products, Basel, Switzerland, in powder format, then encapsulated for the purpose of this trial. Maltodextrin was used as a placebo control.

The results
Fruitflow® significantly decreased fasting blood and urine TMAO when compared to baseline as well as plasma LPS, a marker of intestinal permeability and low-grade inflammation. Moreover, when compared to the placebo group, this was also significantly different for urine TMAO. Dr. Steinert further explained the mechanism of action studied in this trial: Indeed, Fruitflow® seems to modulate the gut microbiota in a way that resulted in a lowering of urine and plasma TMAO. Moreover, we found that Fruitflow® reduced plasma lipopolysaccharides (LPS), a gut microbiota-derived factor that has been suggested to drive onset and progression of chronic inflammation-related diseases such as obesity, type 2 diabetes or non-alcoholic fatty liver disease (NAFLD).
As further mentioned by the authors, a clear distinction was observed also between plasma samples when applying untargeted metabolomics, with TMAO being the top-ranking feature driving the differences found across the Fruitflow® product group compared to the control group. Significant changes in microbial beta diversity were also highlighted. In addition, the microbiome composition changed significantly in the Fruitflow® product group. Lower levels of Bacteroides,Ruminococccus and Hungatella were found, which are known for the involvement in TMA/TMAO metabolism. Dr. Steinert developed on the framework of these results within the overall gut-heart axis research: certainly, these results support earlier findings that polyphenol-rich extracts can lower TMAO concentrations and that this may be related to a targeted beneficial modulation of the gut microbiota. Dr. Claesson added: In this study, we identified set of bacteria that could possibly lower TMAO levels and can potentially be used as a targeted therapeutic strategy for treating cardiovascular disease. It has been proposed that species from the genus Bacteroides can convert TMAO to TMA. We found that the Bacteroides species namely, Bacteroides uniformis and Bacteroides ovatus and Bacteroides acidifaciens, were less abundant in Fruitflow® product group compare to controls explaining observed effect on TMAO via reduction in Bacteroides species. We also noted a significant decrease in Rumonococcus faecis and Hungatella hathewayi within the Fruitflow® product group. It is crucial to remember that Ruminococcus was discovered to have a positive correlation with plasma TMAO levels in a study on rats, while Hungatella hathewayi is known as a TMA producer.
The findings of this study are in line with the studies showing a reduction in plasma TMAO suggesting reproducible effects with polyphenol rich extracts. As a conclusion of this trial we could assert that polyphenol-rich extracts can modulate the gut microbiota conferring a host health benefit.
References and notes
- Fruitflow® is a trademark of Provexis, plc.
- Zhu W, Gregory JC, Org E, Buffa JA, Gupta N, Wang Z, Li L, Fu X, Wu Y, Mehrabian M, Sartor RB, McIntyre TM, Silverstein RL, Tang WHW, DiDonato JA, Brown JM, Lusis AJ, Hazen SL. Gut Microbial Metabolite TMAO Enhances Platelet Hyperreactivity and Thrombosis Risk. Cell. 2016 Mar 24;165(1):111-124. doi: 10.1016/j.cell.2016.02.011. Epub 2016 Mar 10. PMID: 26972052; PMCID: PMC4862743.
- Sandek A, Bauditz J, Swidsinski A, Buhner S, Weber-Eibel J, von Haehling S, Schroedl W, Karhausen T, Doehner W, Rauchhaus M, Poole-Wilson P, Volk HD, Lochs H, Anker SD. Altered intestinal function in patients with chronic heart failure. J Am Coll Cardiol. 2007 Oct 16;50(16):1561-9. doi: 10.1016/j.jacc.2007.07.016. Epub 2007 Oct 1. PMID: 17936155.
- O'Kennedy N, Raederstorff D, Duttaroy AK. Fruitflow®: the first European Food Safety Authority-approved natural cardio-protective functional ingredient. Eur J Nutr. 2017 Mar;56(2):461-482. doi: 10.1007/s00394-016-1265-2. Epub 2016 Jul 7. PMID: 27388464; PMCID: PMC5334395.
Chen H, Zhang S, Wang H, Bao L, Wu W, Qi R. Fruitflow inhibits platelet function by suppressing Akt/GSK3β, Syk/PLCγ2 and p38 MAPK phosphorylation in collagen-stimulated platelets. BMC Complement Med Ther. 2022 Mar 17;22(1):75. doi: 10.1186/s12906-022-03558-5. PMID: 35300669; PMCID: PMC8932123.
Zhang S, Chen H, Li C, Chen B, Gong H, Zhao Y, Qi R. Water-Soluble Tomato Extract Fruitflow Alters the Phosphoproteomic Profile of Collagen-Stimulated Platelets. Front Pharmacol. 2021 Sep 27;12:746107. doi: 10.3389/fphar.2021.746107. PMID: 34646142; PMCID: PMC8502824.
O'Kennedy N, Duss R, Duttaroy AK. Dietary Antiplatelets: A New Perspective on the Health Benefits of the Water-Soluble Tomato Concentrate Fruitflow®. Nutrients. 2021 Jun 25;13(7):2184. doi: 10.3390/nu13072184. PMID: 34201950; PMCID: PMC8308204.
Das RK, Datta T, Biswas D, Duss R, O'Kennedy N, Duttaroy AK. Evaluation of the equivalence of different intakes of Fruitflow in affecting platelet aggregation and thrombin generation capacity in a randomized, double-blinded pilot study in male subjects. BMC Nutr. 2021 Dec 6;7(1):80. doi: 10.1186/s40795-021-00485-5. PMID: 34865648; PMCID: PMC8647412.
Uddin M, Biswas D, Ghosh A, O'Kennedy N, Duttaroy AK. Consumption of Fruitflow® lowers blood pressure in pre-hypertensive males: a randomised, placebo controlled, double blind, cross-over study. Int J Food Sci Nutr. 2018 Jun;69(4):494-502. doi: 10.1080/09637486.2017.1376621. Epub 2017 Sep 18. PMID: 28918674.
Cámara M, Fernández-Ruiz V, Sánchez-Mata MC, Cámara RM, Domínguez L, Sesso HD. Scientific Evidence of the Beneficial Effects of Tomato Products on Cardiovascular Disease and Platelet Aggregation. Front Nutr. 2022 Mar 15;9:849841. doi: 10.3389/fnut.2022.849841. PMID: 35369095; PMCID: PMC8965467.
O'Kennedy N, Crosbie L, Song HJ, Zhang X, Horgan G, Duttaroy AK. A randomised controlled trial comparing a dietary antiplatelet, the water-soluble tomato extract Fruitflow, with 75 mg aspirin in healthy subjects. Eur J Clin Nutr. 2017 Jun;71(6):723-730. doi: 10.1038/ejcn.2016.222. Epub 2016 Nov 23. PMID: 27876806; PMCID: PMC5470100.
Biswas D, Uddin MM, Dizdarevic LL, Jørgensen A, Duttaroy AK. Inhibition of angiotensin-converting enzyme by aqueous extract of tomato. Eur J Nutr. 2014 Dec;53(8):1699-706. doi: 10.1007/s00394-014-0676-1. Epub 2014 Feb 27. PMID: 24573416.
Das D, Adhikary S, Das RK, Banerjee A, Radhakrishnan AK, Paul S, Pathak S, Duttaroy AK. Bioactive food components and their inhibitory actions in multiple platelet pathways. J Food Biochem. 2022 Dec;46(12):e14476. doi: 10.1111/jfbc.14476. Epub 2022 Oct 11. PMID: 36219755.
Schwager J, Richard N, Mussler B, Raederstorff D. Tomato Aqueous Extract Modulates the Inflammatory Profile of Immune Cells and Endothelial Cells. Molecules. 2016 Jan 29;21(2):168. doi: 10.3390/molecules21020168. PMID: 26840280; PMCID: PMC6273921.
Customer’s publication




