Panel discussion on...
Pet Supplements
J.F. (Jake) Burlet, DVM, MBA, ICD.D1
Ronaldo Avella Lavado, BVetMed, MSc2
1. CEO, CanBiocin Inc.
2. Business Development, Operations and Regulatory Affairs Manager, Paul Dick & Associates Ltd. (Pda)
On behalf of International Probiotics Association


Member of AgroFOOD Industry Hi Tech's Scientific Advisory Board


Manufacturing Formulations for Pet Supplements
Contribution by J.F. Burlet
2A) Palatability is a primary challenge: for pet supplements to deliver a benefit, the first hurdle is to ensure the supplement is willingly consumed. Felines are notoriously picky. Stability is equally complex, especially for probiotics, omega-3s, and bioactives which can be sensitive to heat, moisture, and oxidation. When blending multiple ingredients into a final supplement formulation, avoiding unintentional incompatibilities between ingredients is key. Ensuring even distribution of actives in final dose delivery forms (chews, gummies, powders, etc.) is important for efficacy. As pets vary widely in size and metabolism, achieving dosing accuracy is important.
2B) Ultimately, the formulation choice best considers the target species, individual pet preferences, and ease of use for the pet owner to ensure consistent and compliant usage. Chews are often preferred due to convenience and being easy to incorporate into daily routines. Powders offer flexibility, but are dependent on food acceptance and accurate measurement by owners. Convenience, ease of administration, and clear dosing instructions determine whether products are used consistently enough to deliver benefits to the animal (and pet owner)!
2C) Multi-active formulations remain market leading as many pet health concerns are multifactorial and benefit from synergistic mechanisms. It remains important to recognize and acknowledge that in many instances we “don’t know that we don’t know”. The intestinal microbiome is an incredibly diverse and complex environment. Pet supplements most certainly have the ability to effect change within and upon the pet microbiome. While some of these effects are known and documented, we are only just beginning to learn more about metabolomics and specific metabolites which can better explain mechanisms of action in pets. It remains an exciting time for pet owners, suppliers of animal wellness formulations and most importantly pets, as multi-active formulations continue to demonstrate that pet healthspan and lifespan can be positively influenced. Formulating with known prebiotic, probiotic and postbiotic bioactives (collectively synbiotics) can deliver better functional outcomes for companion animals.
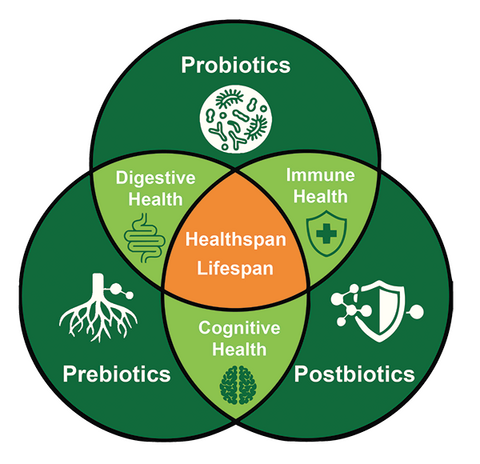
2D) One cannot simply assume that all biotics function identically between pet species. Species-specific anatomy and physiology significantly informs and guides formulation. For example, dogs and cats have notable differences between the binding ability of probiotics to intestinal epithelial cells. Dogs have a longer and more complex gastrointestinal tract compared to cats. Intestinal pH and transit times are different. Cats are obligate carnivores and have different protein and amino acid needs compared to dogs. Smaller pets may require lower doses or different delivery forms. Age and stage of life define that growing puppies and kittens, adult pets, and senior animals each have unique nutritional needs to be considered. Tailored formulations help meet the diverse needs of the pet population; one size does not fit all.
4A) Scientific substantiation drives credibility. Products supported by species-specific trials, transparent labeling, and demonstrated bioavailability will differentiate themselves from generic marketplace offerings. Regulatory convergence and clearer guidance will enhance consistency across markets. Quality assurance and quality control (GMP compliance, contaminant testing, and traceable sourcing) are drivers of formulation reputation of brands. Much can also be learned and leveraged from work being conducted in the fields of human and environmental health (transferable “One Health” fundamentals). We are better when we work together to improve the health of animals, people and the environment.
4B) When suppliers, formulators, veterinarians, pet owners, distributors and retailers can more easily verify that a product’s dosage and benefits are grounded in credible research, confidence will increase substantially. Transparency, rather than marketing intensity, will foster trust and sustainable growth. Widespread adoption of minimum evidence standards, backing efficacy claims, will help drive the industry forward in a sustainable and responsible manner.
Current State and Future Directions of Companion Animals Supplement Regulation
Contribution by Ronaldo Avella Lavado
Regulation of Pet Supplements in the United States and Europe
Pet supplements are subject to regulatory oversight designed to ensure high standards of quality, safety, and efficacy. While regulatory frameworks differ between jurisdictions, both the United States and Europe require adherence to good manufacturing practices (GMPs), appropriate documentation for pre-market authorisation, and compliance with rules governing ingredients, labeling, and permitted claims.
In the United States, the National Animal Supplement Council (NASC) plays a key role in promoting quality and post-market vigilance within the animal supplement industry. NASC establishes standards for its members, including manufacturing guidelines, adverse event reporting systems, and label review processes.
In Europe, regulatory oversight involves both legal authorities and industry organizations that contribute to compliance and best practices. The European Pet Food Industry Federation (FEDIAF) provides guidance on labeling, nutritional standards, and responsible marketing. The European Federation of Specialty Feed Ingredients (FEFANA) represents producers of specialty feed ingredients such as vitamins, amino acids, enzymes, probiotics, and other functional additives, supporting regulatory alignment and product safety.
Regulatory frameworks for “biotics” vary across jurisdictions. In the United States, these products are referred to as “Direct-Fed Microbials” and are defined as “a source of live (viable), naturally occurring microorganisms.” In contrast, Europe uses the term “Gut Flora Stabilizers,” classified within the functional group of zootechnical feed additives and defined as “microorganisms or other chemically defined substances which, when fed to animals, have a positive effect on the gut flora.”
Labelling and Claims Are Critical and Challenging
Clear and compliant labeling drives success in the pet supplement market. Pet supplements must demonstrate safety, quality, and intended use, but they can only make structure or function claims. Acceptable language includes promote, support, or maintain normal health or physiological function. If a product uses drug-like or therapeutic claims, regulators may reclassify it as a pharmaceutical. Other risks include using prescription-only ingredients or implying disease outcomes without authorization.
Regulatory oversight will likely increase as demand grows for preventive and wellness solutions. Authorities may strengthen transparency and evidence standards while aiming for greater alignment across jurisdictions. This trend reflects a more informed consumer base and a more structured global marketplace.
Veterinarians and Their Influence on Industry and Consumer Relationships
Veterinarians play a central role in the pet supplement market, bridging science, industry, and pet owners. Their clinical expertise supports responsible product development, evaluation of ingredient functionality, and assessment of compliant, evidence-based claims.
Strengthening collaboration among veterinarians, consumers, and industry stakeholders is essential for continued growth. Greater education, transparent communication, and veterinary involvement in product development can enhance trust, ensure safety, and support regulatory compliance across the sector.
Future Perspectives of the Pet Supplements Industry
As the pet supplements market continues to expand, stronger regulatory clarity and higher quality standards will be essential. Clearer labeling guidelines and more precise requirements for claim development are needed to ensure transparency and prevent misleading communication. Claims should be firmly grounded in scientific research, supporting both product safety and demonstrated efficacy. Importantly, regulatory consultants will play a key role in supporting product development, regulatory submissions, and market entry.
Strengthening GMPs is essential to improve product consistency and reliability in the pet supplement market. Continued support for industry oversight bodies such as the NASC will further enhance quality assurance and post market monitoring. Although the market is expected to grow, long term success will depend on improving safety, quality, and scientific credibility. Greater collaboration among pet owners, veterinarians, and industry stakeholders will support responsible innovation and improve outcomes for pets and their families.
What single change—scientific, regulatory, or educational—would most improve consumer trust in PET supplements over the next decade?
An impactful change to improve consumer trust in pet supplements over the next decade would be the implementation of harmonized regulatory standards that require both rigorous quality control and substantiated health claims. In this context, regulatory consultants will play a critical role in guiding product development, managing regulatory submissions, and facilitating market entry, all grounded in scientific evidence.
In this Panel Discussion, several prominent companies within the food and nutraceutical ingredient industry have been invited to discuss about drivers and barriers of healthy lifestyle, focusing on global and regional consumer trends, scientific achievements, emerging delivery formats, use of AI technologies and the implementation of the United Nations sustainability goals.
References and notes
- Schunck, M. , Louton, H. and Oesser, S. (2017) The Effectiveness of Specific Collagen Peptides on Osteoarthritis in Dogs-Impact on Metabolic Processes in Canine Chondrocytes. Open Journal of Animal Sciences, 7, 254-266. doi: 10.4236/ojas.2017.73020.
- Koivisto et al., 2014; Siebert et al., 2010
- The oral intake of specific Bioactive Collagen Peptides (BCP) improves gait and quality of life in canine osteoarthritis patients—A translational large animal model for a nutritional therapy option
Dobenecker B, Böswald LF, Reese S, Steigmeier-Raith S, Trillig L, et al. (2024) The oral intake of specific Bioactive Collagen Peptides (BCP) improves gait and quality of life in canine osteoarthritis patients—A translational large animal model for a nutritional therapy option. PLOS ONE 19(9): e0308378. https://doi.org/10.1371/journal.pone.0308378
Questions
1.
2.
3.
4.
Ingredients companies - clinical data
A) What are the main scientific gaps that still exist in PET supplement development compared to human dietary supplements?
B) How important is species-specific research when selecting and developing active ingredients for pets? Can human data ever be sufficient?
C) Is the humanization of pets helping consumers make better-informed choices, or does it risk creating unrealistic expectations about supplement performance?
D) From an ingredient supplier’s perspective, which quality parameters (standardization, bioavailability, purity) are most critical for PET applications?
E) How are trends such as “clean label,” sustainability, and transparency influencing pet owners’ purchasing decisions in the PET supplement space?
F) What type of clinical evidence should realistically be expected to support PET supplement claims today?
G) Which types of claims are most likely to be misunderstood by consumers, and how can this risk be reduced through clearer substantiation and labeling?
H) What are the main methodological challenges in conducting clinical trials for companion animals, and how can they be addressed?
I) How reliable are owner-reported outcomes compared to veterinary assessments, and how should they be integrated into study design?
L) Have you noticed an increasing trend in the use of one (or more) ingredients for pet supplements formulated to promote healthy ageing?
Formulation
A) What are the biggest formulation challenges in PET supplements, particularly regarding palatability, stability, and dosing accuracy?
B) How do formulation choices (e.g., chews, powders, liquids) influence compliance and consistent use from a consumer perspective?
C) Do you see a shift toward simpler, single-ingredient formulations, or are multi-active blends still the dominant approach? Why?
D) How do species differences (dogs vs cats, size, age) influence formulation strategies?
E) Omega 3 alternatives for pet nutrition and sustainability: how do the innovative omega-3s for pet food stack up against their traditional fishy counterparts?
Regulation
A) How do regulatory frameworks for PET supplements differ between the EU and the US, and what challenges do these differences create for global brands?
B) Which types of claims represent the highest regulatory risk today, and which are more likely to be acceptable if properly substantiated?
C) Do you expect regulatory oversight of PET supplements to become stricter in the coming years? Why or why not?
D) What role should veterinarians play in guiding pet owners’ choices regarding PET supplements, and how can trust between brands, vets, and consumers be strengthened?
Open questions
A) Looking ahead 5–10 years, what will be the key factors determining the credibility and long-term success of the PET supplements sector?
B) In your view, what single change—scientific, regulatory, or educational—would most improve consumer trust in PET supplements over the next decade?
References and notes
Panelists

















































