UNDERSTANDING THE RELATIONSHIPS BETWEEN DIET, MICROBIOME AND METABOLISM


Almost 10 years ago, a major study (1) changed how we look at weight. It involved genetically identical twins discordant for obesity who both provided fecal samples. Their microbial communities were then transferred to germ-free mice, and the mice who received the microbiota from obese participants grew fat, while the recipients of lean participants stayed lean, as illustrated above.
This was one of the first demonstrations of a cause-to-effect relationship between the microbiota and weight.
Now, starting 2022, what else have we learned about the microbiome in relation to weight regulation? Here are three focuses to better understand the relationship between diet, microbiome, and metabolic health.
INTRODUCTION
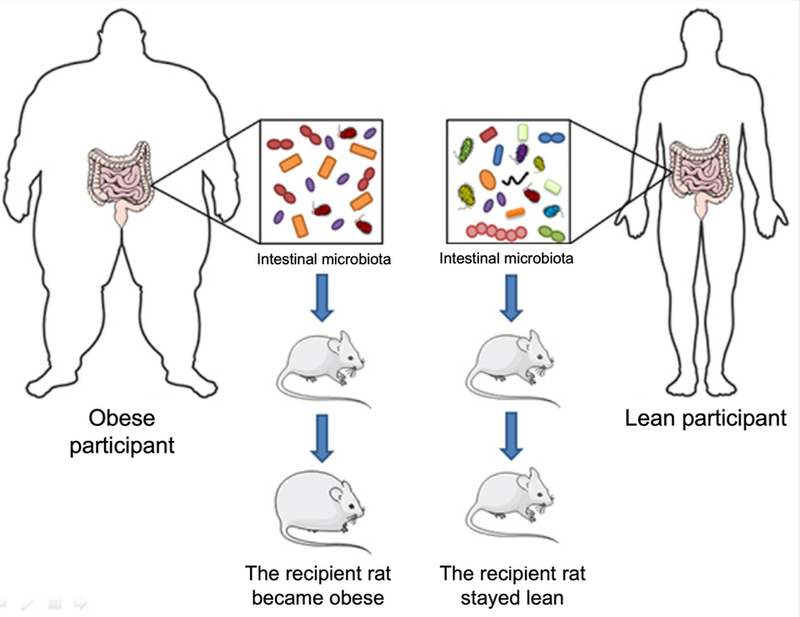
Image from Wikimedia adapted by Author
When you eat a high-fat diet rich in animal products such as meat, cheese and eggs, you bring in choline, phosphatidylcholine and L-carnitine. These compounds are not a problem per se, but when they go through the process of digestion and are processed by the gut microbiota they are transformed into trimethylamine (TMA). In turn, trimethylamine in the liver is oxidized into trimethylamine-N-oxide (TMAO), a compound known for its toxicity and proatherogenic property: it increases risks of arteria hardening, clogs and cardiovascular disease - Zhu et al., 2020 (5).
Studies have determined which are the microbial culprits (7) responsible for these metabolic conversions and even proposed probiotics (8) to reduce these bacteria and their pathways.
However, what seems the simplest, knowing the food sources causing the rupture in balance, would be to adapt the diet accordingly. If the bacteria thriving on these foods no longer receive substantial amounts of nutrients, their abundance will naturally decrease, and they will be less numerous to transform a weekly rib-eye into stiff arteries.
Reducing red meat, liver, poultry, fish, milk, and cheese in favor of more plant-based foods brings benefits to the microbiota and cardiovascular health and is more sustainable (9).
TMAO: WHAT YOU EAT IS WHAT YOUR GUT BACTERIA EAT — FOR BETTER OR FOR WORSE
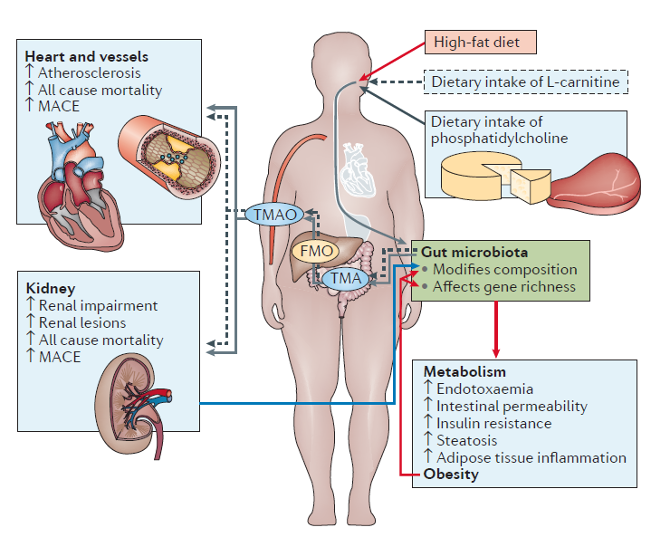
Diet, microbiota, TMAO and health outcomes, from a review by Aron-Wisnewsky and Clément (6)
New studies conducted with state-of-the-art sequencing methods can identify microbial markers in fecal samples down to the genes expressed and metabolites produced by bacteria. Such a study (10) recently found a negative correlation between the abundance of bacterial chaperone metabolite Caseinolytic peptidase B (ClpB) and BMI. In a cohort of 131 subjects, the leaner the participants, the more ClpB was present in their feces.
ClpB has been described as a conformational antigen mimetic of alpha-MSH (11) one of the key hormones regulating appetite. As ClpB presents a homology of sequence and structure to alpha-MSH, it is able to bind to the hormone receptors (12) and activate their answer, which up-signals feeling of fullness.
We don’t know yet the chicken from the egg: will a reduction in food intake increase levels of ClpB-producing bacteria in the gut? For now we don’t know, but the cause to effect relationship has been demonstrated in mice studies (13, 14) and in overweight adults (15) confirming that the intake of a specific probiotic from French cheese, Hafnia alvei HA4597 that produces ClpB, reduces appetite, food intake, and supports weight loss.
ClpB: A MICROBIAL METABOLITE SLIPS INTO HORMONAL REGULATION OF APPETITE
Diet impacts the microbiota, which in turn impacts health status, metabolism and weight. There’s a good chance that the diet acts as a double sword: eat well, and the microbiota will exert protective effects. But eat too much food, too much fat, too many animal products and insufficient fiber and a deleterious microbiota will develop which will further impact your weight gain, atherosclerosis and even possibly increase your appetite.
This is empowering and gives room to optimism, because studies show a change in diet has an almost immediate impact (16) on the microbiota - although it can take about a year to normalize levels of Firmicutes/Bacteroidetes (4). Basically, going on a diet, or healthier eating habits, can empower the microbial community to these efforts and make them easier and more fruitful.
CONCLUSION
Nina Vinot has a background as a life sciences engineer from AgroParisTech and conducted nutrition research at Penn State University, CNRS and INRAe.
She has now 10 years experience promoting the use of beneficial bacteria, a track record of success developing sales across Europe for Probiotical, and is today international sales director for the French biotech TargEDys, pioneering precision probiotics.

Many studies comparing the microbiota of obese and lean people conclude that obese people are characterized by a lower biodiversity and a higher Firmicutes/Bacteroidetes ratio. Without going into depths about these two groups of microorganisms that represent 90-95% of the gut inhabitants, what is important to understand is that Firmicutes are able to harvest more energy from food and deliver the host more energy following their own degradation of the nutrients in the gut.
Alanna Collen in her book 10% Human (2) explained the difference accounts for roughly 2% more calories absorbed per day (40 to 50 calories for an average diet). A quick calculation means a lean person would put on 1 kg every 5 months. At this rhythm, a healthy weight becomes overweight in 5 years and obese in 10, just because of the microbiota markup.
And this could even be underestimated. A magnificent study published in 2011 (3) looking precisely at the calories in and out estimates that for each increase of 20% in Firmicutes, the additional energy harvest amounts to 150 calories per day!
Why is this important?
Because the diet influences this Firmicutes/Bacteroidetes ratio. In an intervention study back in 2006 (4) 12 obese people on two different diets were monitored to look at the diet’s effect on their microbiota: little by little, the ratio normalized towards the profile typical of lean individuals.
FIRMICUTES/BACTEROIDETES: HOW TO INTERPRET THIS INDEX
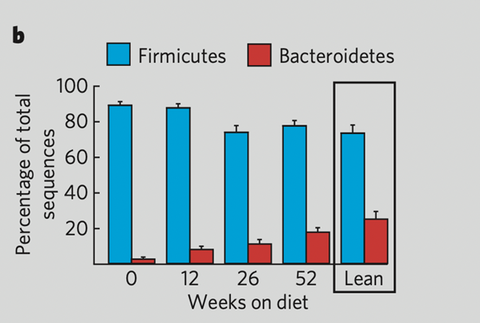
Evolution of the Firmicutes and Bacteroidetes percentages during low fat and low-carb diets, from Nature Brief Communications
For health practitioners, dietitians, nutritionists, it also means that knowing patients Firmicutes/Bacteroidetes ratio will help tailor the daily caloric needs. With 30% more Firmicutes, the daily energy needs can be reduced by 225 calories.
How does the diet regulate Firmicutes/Bacteroidetes?
Firmicutes love carbohydrates when they are abundant, while Bacteroidetes love carbohydrates when there’s not too much. That’s simply because the bacterial output from digesting these sugars and fibers are fatty acids that increase local acidity. Firmicutes are tolerant to acidity, while Bacteroidetes will sense pH reduction and will stop eating to preserve themselves. In consequence, they will leave some food undigested, and the host will excrete more unharvested energy.
In consequence, the more we eat, the more we favor Firmicutes to the detriment of Bacteroidetes, and the more they will harvest energy from our food intake. It’s a vicious cycle.
MICROBIOME




